Advil 12 Hour for fast and extended acute pain relief, up to 12 hours1
Advil 12 Hour’s dual-layer technology ensures fast, and extended, acute pain relief1
Designed with dual-layer technology for up to 12 hours of pain relief with one pill. A quick burst for relief that’s fast. Extended release for relief that lasts.1
A more convenient option for patients — one Advil 12 Hour* provides the same amount of medication as 200 mg of standard ibuprofen given every 4 hours.1
* Ibuprofen is the nonsteroidal anti-inflammatory ingredient in Advil products.1
Advil contains ibuprofen – a nonsteroidal anti-inflammatory drug (NSAID)
Ibuprofen is the active ingredient in Advil and Children’s Advil that relieves pain and reduces fever. Like all NSAIDs, ibuprofen is an analgesic, antipyretic, and anti-inflammatory.1
NSAID = nonsteroidal anti-inflammatory drug.

Overall & GI Tolerability Profile
Ibuprofen comparative tolerability in a large-scale randomized trial
In the PAIN study, published in Clinical Drug Investigation, and which included over 8,500 patients, overall tolerability of ibuprofen was:1,2
- Statistically equivalent to that of acetaminophen
- Superior to that of ASA
This large scale randomized trial comparing nonprescription doses of ASA, acetaminophen, and ibuprofen in 8,677 adults measured rates of significant adverse events related to tolerability. The primary outcome measure was the number of patients with at least one significant adverse event, defined as an event that was serious, severe or moderate, resulted in a second physician consultation, led to cessation of treatment, or was of missing intensity. Statistical analysis tested for equivalence between ibuprofen and acetaminophen and for difference with ASA.1,2*
ASA = acetylsalicylic acid; GI = gastrointestinal.* This was a blinded, multicentre study in general practice of up to 7 days of ASA, acetaminophen (both up to 3 g daily) or ibuprofen (up to 1.2 g daily), administered for common painful conditions, using patient generated data with physician assistance. 1,108 general practitioners included 8,677 adults (2,900 ASA; 2,886 ibuprofen; 2,888 acetaminophen; 3 patients had no code label number). The main indications were musculoskeletal or back pain (48%), sore throat, the common cold and flu (31%).

When used as directed for acute pain, OTC ibuprofen is well tolerated1*
In the PAIN study, which included over 8,500 patients, total GI events (including dyspepsia) and abdominal pain were less frequent with ibuprofen (200 mg) compared to ASA (500 mg) or acetaminophen (500 mg) (all p<0.035).1,2
This large-scale randomized trial comparing nonprescription doses of ASA, acetaminophen, and ibuprofen in 8,677 adults measured rates of significant adverse events related to tolerability. The primary outcome measure was the number of patients with at least one significant adverse event, defined as an event that was serious, severe or moderate, resulted in a second physician consultation, led to cessation of treatment, or was of missing intensity. Statistical analysis tested for equivalence between ibuprofen and acetaminophen, and for difference with ASA.1,2*
ASA = acetylsalicylic acid; GI = gastrointestinal; OTC = over the counter.* This was a blinded, multicentre study in general practice of up to 7 days of aspirin, acetaminophen (both up to 3 g daily), or ibuprofen (up to 1.2 g daily), administered for common painful conditions, using patient generated data with physician assistance. 1,108 general practitioners included 8,677 adults (2,900 aspirin; 2,886 ibuprofen; 2,888 paracetamol; 3 patients had no code label number). The main indications were musculoskeletal or back pain (48%), sore throat, the common cold and flu (31%).
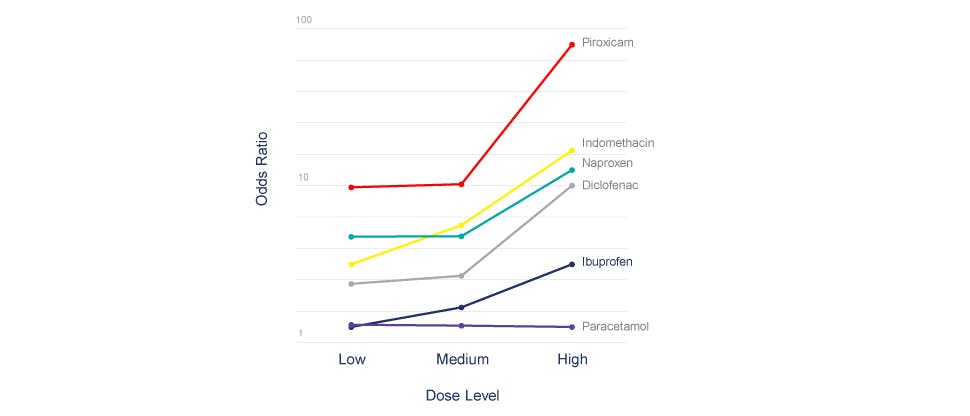
Dose-response relationships for the risks (odds ratios) of upper GI bleeding with individual NANSAIDs
The chart above demonstrates the dose response relationship for the risks (odds ratios) of upper GI bleeding with individual NSAIDs.
In the meta-analysis, ibuprofen at a low dose (<1200 mg) had an odds ratio (OR) comparable to acetaminophen and lower than other NSAIDs. Even at medium doses (1200–1799 mg), ibuprofen showed a lower OR (1.8) for the risk of serious upper GI bleeding compared to other NSAIDs, followed by diclofenac (3.2), naproxen (5.4) and indomethacin (6.8).3*†
The meta analysis included individual patient data from three retrospective case control studies using similar data collection protocols, the main outcome measures were risks associated with individual NANSAIDs according to dose used and the period of time for which they were given.
CI = confidence interval; GI = gastrointestinal; NANSAID = non aspirin nonsteroidal anti-inflammatory drugs; NSAID = nonsteroidal anti-inflammatory drug; UGI = upper gastrointestinal.* Odds ratios were relative to non-use of any NANSAID or aspirin, and were adjusted for use of aspirin, use of anticoagulants, smoking, and history of UGI problems.† Ibuprofen showed the lowest odds ratio (OR = 1.7; 95% CI: 1.1, 2.5), followed by diclofenac (4.9; 95% CI: 3.3, 7.1), indomethacin (6.0; 95% CI: 3.6, 10.0), naproxen (9.1; 95% CI: 6.0 13.7), piroxicam (13.1; 95% CI: 7.9 21.8) and ketoprofen (34.9; 95% CI: 12.7, 96.5). The meta-analysis was carried out in hospitals in Catalonia, England, Scotland, and Sweden. There were 2,472 cases of UGI bleeding and 5,877 controls that were studied.
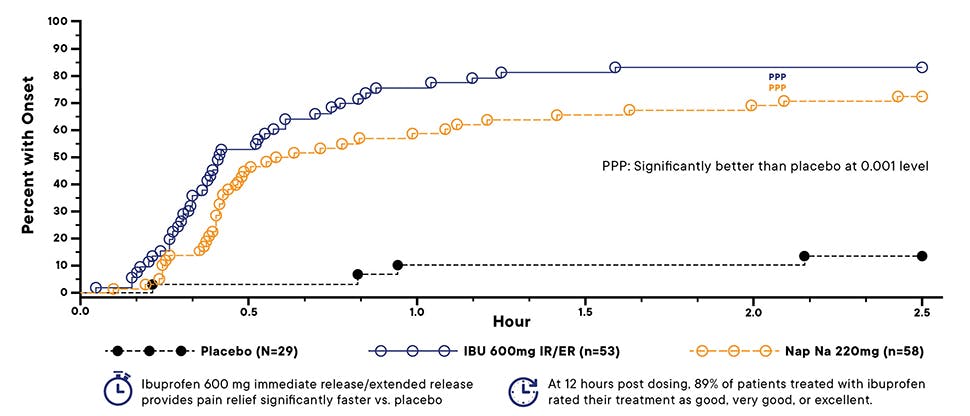
Clinically proven efficacy in headache pain
Data from Christensen et al. (ibuprofen 600 mg immediate release/extended release) demonstrated: 4,5*
- Rapid onset of analgesia with a significantly higher percentage of subjects experiencing first perceptible relief with ibuprofen vs. placebo, as early as 25 minutes to 30 minutes (p<0.001) after the dose was taken
- Ibuprofen provided significantly better pain relief over 12 hours vs. placebo (p<0.001)
*Christensen et al. evaluated the efficacy and safety of ibuprofen 600 mg immediate release/extended release caplets. This study demonstrated that ibuprofen 600 mg immediate release/extended release provides a rapid onset of analgesia that was maintained for at least 12 hours.

Clinically proven analgesia and relief from headache pain
Advil (200 mg, 400 mg) has been clinically proven to effectively relieve headache pain.1
Journal of Clinical Pharmacology, 1996 (Schachtel BP, et al.)
- A double blind, randomized study comparing ibuprofen 400 mg (N=153) vs. acetaminophen 1000 mg (N=151) and placebo (N=151) found that ibuprofen relieved headache pain significantly better than acetaminophen 1000 mg and placebo. Participants receiving ibuprofen 400 mg reported experiencing no pain in a significantly faster (at 1, 2, 3, and 4 hours post intake) than those receiving acetaminophen 1000 mg. Approximately, more than 3 times the percentage of subjects receiving ibuprofen 400 mg noted no pain compared to acetaminophen 1000 mg 3 hours post dose.6*
Headache, 1988 (Schachtel BP, et al.)
- A double blind, randomized study compared ibuprofen 400 mg (N=35) to placebo demonstrating significant analgesic effect on headache within 30 minutes (N=35)7†
* At 30 minutes and 1, 2, 3, and 4 hours post dose, each patient completed the Headache Pain Intensity Scale and the Headache Pain Relief Scale.† Seventy subjects satisfied the admission criteria and included men (21 in treatment group and 15 in placebo) and women (15 in the treatment group and 20 in the placebo group) with a mean age of ~21 years of age. They were instructed to complete a Headache Diary when they experienced a muscle contraction headache. When the headache occurred, they were to swallow a single dose of study medication. Efficacy evaluations were done at 15, 30, 45, 60, 90, and 120 minutes after dosing.

Clinically proven efficacy in migraine pain1,8
Ibuprofen 400 mg has been clinically proven to be effective in relieving the pain of migraine headache and related functional disability.8*
Cochrane Database of Systematic Reviews, 2013 (Rabbie R, et al.)
- In a review of 9 studies all participants had a diagnosis of migraines and most had a history of migraine symptoms for at least 12 months before entering the study
- The proportion of participants experiencing headache relief at 2 hours with ibuprofen 400 mg was 57% vs. 25% with placebo8†
- The proportion of participants with relief from functional disability at 2 hours after ibuprofen 400 mg was 42% vs. 24% with placebo8‡
NNT = number needed to treat.* Included participants all had a diagnosis of migraine headaches according to International Headache Society criteria. The mean age of participants was 30 to 40 years of age in individual studies.
† Proportion with ibuprofen is 57% (528/931; range 41% to 72%) and the proportion with placebo was 25% (224/884; range 7% to 50%).
‡ The proportion of participants with relief of functional disability at 2 hours after ibuprofen 400 mg was 42% (245/583; range 18% to 76%). The proportion of participants with relief of functional disability at 2 hours after placebo was 24% (129/531; range 13% to 55%).

Advil 12 Hour is an ideal option for Ellen’s tension headaches
Ellen works long hours as a middle school teacher and describes herself as ‘stressed out’ yet committed to her students. She wants to avoid missing teaching days and looking for effective relief that will work fast and last.
Advil 12 Hour:1
- Has been proven to effectively relieve headache and migraine pain
- Provides rapid onset of analgesia for up to 12 hours
OTC ibuprofen has a well-established safety profile when used as directed.
OTC = over the counter.* Fictional case study.
Recommend Advil to manage your patients’ acute pain
Advil 12 Hour
Advil 12 Hour’s dual-layer technology ensures fast, and extended, acute pain relief1
Advil — for all your patients’ acute pain relief needs

Advil — for all your patients’ acute pain relief needs
Learn more about the Advil product family range.
