Calpol Paediatric suspension

Tough** on fever and pain,1 gentle 1,3 on the child†
Calpol Paediatric suspension contains paracetamol, which is recommended as first-line treatment for fever and mild-to-moderate pain in children.2-4 It starts to relieve fever within 15 minutes of dosing.1,5-7
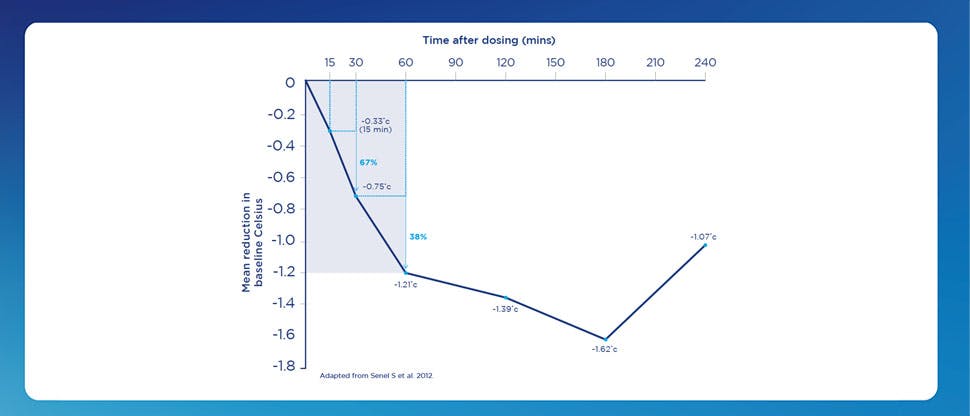
This was a randomised, single dose, open labelled trial involving 316 children (between the ages of 6 months and 12 years) of which 158 were given acetaminophen.
The maximum decrease in temperature occurred 3 hours after administration of paracetamol, however after 30 and 60 mins, there were temperature reductions of 0.75ºC and 1.21ºC.5
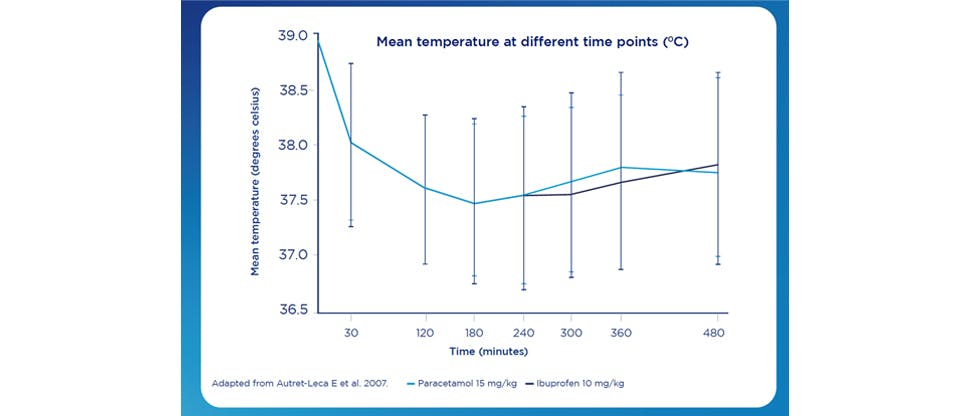
As effective as Ibuprofen at doses of 10 mg/Kg9
This was a multicentre, double-blind, double dummy of 304 patients between the ages of 3 months to 12 years, requiring treatment on an outpatient basis and with a tympanic temperature of at least 38.5°C with an upper boundary of 40.5°C, associated with various pathologies such as sore throat, influenza, respiratory tract infection, ear infection or immunisation.11
Paracetamol was found to be as effective as ibuprofen in treating fever in children, with no significant difference between paracetamol and ibuprofen seen in temperature reduction from baseline up to 6 hours.11

Calpol Paediatric suspension relieves fever and pain and starts working to in 15 minutes.1. As effective as ibuprofen,8 nothing works faster on pain and fever.
It also doesn’t interfere with the stomach’s protective lining.9. Calpol is the No.1 pain relief brand*# with 65 years of experience and expertise and trusted by millions of consumers and HCPs in pain and fever.10
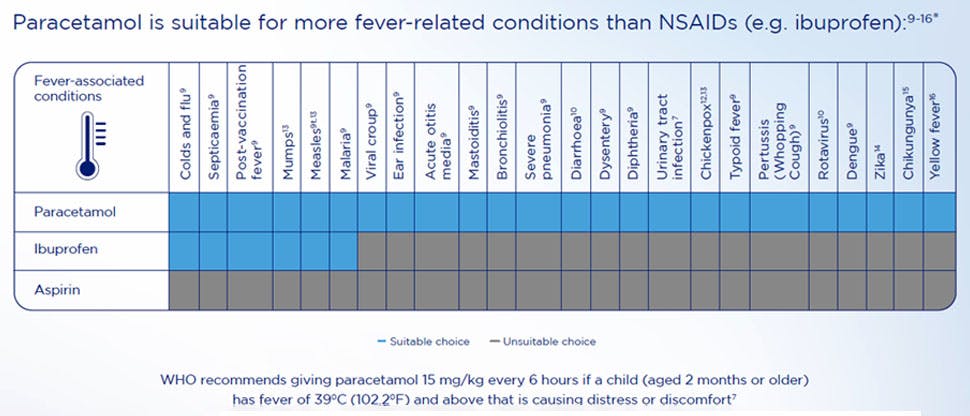
Paracetamol is better suited for children’s pain relief than NSAIDs
Paracetamol is recommended as a first-line treatment for fever and mild-to-moderate pain in children.3,13–15 It is more suitable than NSAIDs such as ibuprofen for fever-related conditions.
Paracetamol is also the medicine of choice for mild pain,12 and is as effective as NSAIDs in pain reduction for migraine headaches,16,17 sore throats,18,19 earache,20,21 teething22,23 and more.24–27
Effective as pain-relief in children28
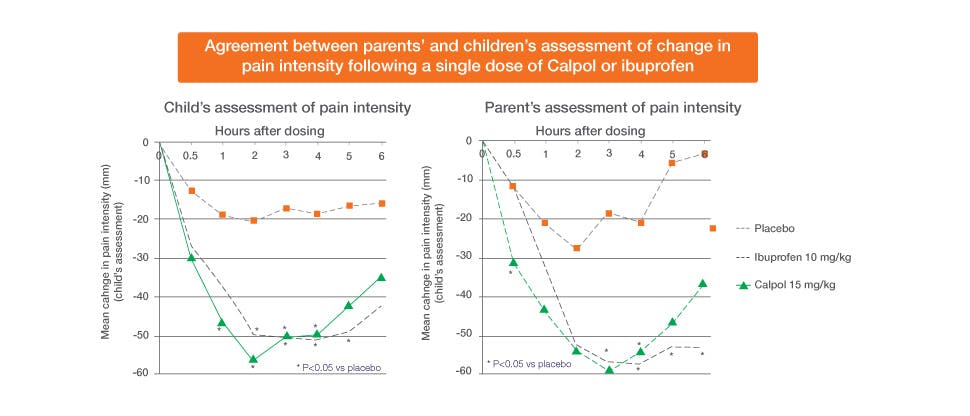
This was a randomised, single-dose, double-blind, placebo-controlled study in children aged 2-12 years with acute sore throat treated with paracetamol 15 mg/kg, ibuprofen 10 mg/kg or placebo. Paracetamol showed significant pain relief vs. placebo from 1-hour post dose, which was sustained until 4 hours after dosing based on the children’s assessment of their pain. The parents’ assessment of changes in pain intensity seen in their children reflected the same.29

When compared to non-steroidal anti-inflammatory drugs (NSAIDs) e.g. ibuprofen, paracetamol is preferred because it is gentle on tiny tummies,1,3 has a lower risk of bleeding complications in certain vector-borne diseases like dengue2 may be suitable for children with dehydration28 and does not interact with as many other medicines like ibuprofen does.2
Learn more

Managing children’s fever and pain
Tailor made for pharmacists, this tool is an excellent guide to help you manage children with fever or pain.

When to refer children with fever
Learn more about “red-flag” symptoms when children present with fever.

Calpol Optizorb technology
Discover more about this revolutionary technology that allows delivery of paracetamol fast5 and effectively6 when it is needed most.