FLONASE Nasal Efficacy

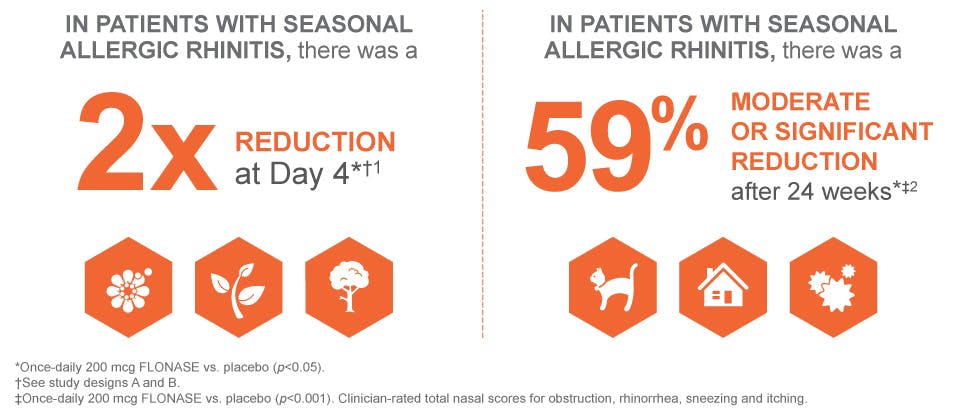
Sustained relief of nasal symptoms in 2 studies of FLONASE vs placebo1,2
Read more about Flonase products

FLONASE SENSIMIST Allergy Relief
Find out how FLONASE SENSIMIST Allergy Relief can help your patients.

Children’s FLONASE SENSIMIST Allergy Relief
Find out how Children’s FLONASE SENSIMIST Allergy Relief can help your patients.

Children’s FLONASE Allergy Relief
Find out how Children’s FLONASE Allergy Relief can help your patients.



